| English Spanish | |
|
|
|
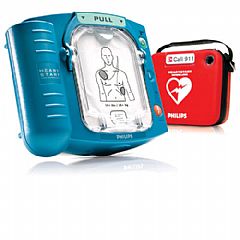
| Philips HeartStart AED Descripción Modelo: OnSite Solicitar Información | ||||
| Imagen | ||||
| ||||
Parte posteriora |
|
7874 NW 64th Street, Miami, Florida, 33166 Tel: (305) 594-0000 - Fax: (305) 594-2020 - © Copyright 2007 - Life Medical Equipment International Inc. |