|
|
Philips HeartStart AED Product Detail
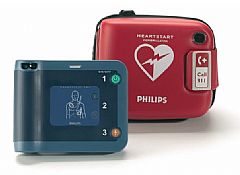
Model: FRx
The Philips HeartStart FRx Defibrillator is designed to be easy to use, rugged and reliable for those who get their first. On the scene with law enforcement, on the field with student athletes or on the job with employees, the Philips FRx defibrillator is the solution for treating SCA from ventricular fibrillation in environments and conditions too demanding for many other defibrillators.
SMART Technology, only from Philips
At the core of all HeartStart Defibrillators is SMART Biphasic technology. The HeartStart FRx Defibrillators biphasic waveform delivers a highly effective defibrillation shock that is also gentle to the heart. No other external defibrillation therapy has been supported by more published clinical data.
SMART Analysis, a proven Philips technology for heart rhythm assessment, ensures that FRx Defibrillator only shocks when it should. SMART Analysis automatically assesses the victims heart rhythm and is designed not to deliver a shock unless the rhythm is determined to be shockable even if the shock button is pressed. Whats more, SMART Pads II enable the FRx to keep pace with the responder by adjusting to their actions.
Designed for real world use
The Philips HeartStart FRx Defibrillator is exceptionally rugged. Designed to surpass rigorous testing requirements, the FRx withstands jetting water, loads up to 500 pounds and a one-meter drop onto concrete.
Built on a platform of proven ease-of-use - The HeartStart FRx Defibrillator was designed to be as easy to use as the HeartStart OnSite Defibrillator and shares many of its features, including CPR Coaching and intuitive icon-driven operation. Although small and lightweight - just 3.5 pounds - the FRx is equipped to guide you through the resuscitation of a SCA victim.
The HeartStart FRx guides you through every step with clear, calm voice commands and descriptive visual icons.
The FRx even reminds you to call emergency medical services (EMS).
Pressing the blue i-button activates HeartStart CPR Coaching for assistance with CPR. The flashing icons and the Quick Reference Guide can be used to lead you through the defibrillation steps - even in situations where hearing voice instructions is a challenge.
Once EMS arrives, hand-off is fast and easy because the FRx is compatible with advanced defibrillators like the HeartStart MRx. With HeartStart adapters, our pads can be plugged into devices from other manufacturers to ensure continuity of care.
Provides a complete, reliable solution with reliability backed by Philips
The HeartStart FRx Defibrillator is powered by an easy to install, long-life (four-year) battery, so you know the device is charged and ready. The device's automated daily, weekly and monthly self-tests check the pads readiness, and verify functionality and calibration of circuits and systems. With over 85 tests, the FRx is one of the most comprehensive self-testing devices on the market and is virtually maintenance-free. The blinking green Ready light on the defibrillator is your assurance that the device is ready for use.
Specifications:
- Defibrillator
- Waveform: Truncated Exponential Biphasic. Waveform parameters adjusted as a function of each patients impedance.
- Energy
- Single energy output:
- Adult: nominal 150 Joules into a 50-ohm load
- Infant/Child: nominal 50 Joules into a 50-ohm load.
- Protocol: Device follows preconfigured settings. Defibrillation and CPR protocol can be customized using HeartStart Event Review or HeartStart Configure Software.
- User Interface
- Instructions: Detailed voice prompts and visual icons guide responder through use of the defibrillator.
- CPR Coaching: Voice coaching for adult and infant/child CPR provides instructions and audio cues for each breath, as well as the appropriate number, rate and depth of chest compressions.
- Controls: Green on/off button, blue i-button, orange shock button, optional infant/child key.
- Indicators: Ready light, blue i-button, caution light, illuminated pads, icons, shock button lights up when shock is advised.
- Physical
- Size: 2.4 x 7.1 x 8.9 inches (6 x 18 x 22 cm) H x D x W.
- Weight: With battery and pads case: 3.5 lbs. (1.5 kg). Without battery or pads case: 2.6 lbs. (1.2 kg).
- Environmental
- Sealing: Waterjet proof IPX5 per IEC60529. Dust protected IP5X per IEC60529.
- Temperature: Operating/Standby: 32° - 122° F (0° - 50° C).
- Altitude: 0 to 15,000 feet.
- Aircraft: Device: RTCA/DO-160D;1997.
- Crush: 500 pounds.
- Vibration: Operating: meets MILSTD 810F Fig.514.5C-17, random; Standby: meets MILSTD 810F Fig.514.5C-18, swept sine.
- EMI (Radiated/Immunity): CISPR II Group I Class B, IEC 61000-4-3, and IEC 61000-4-8.
- Patient Analysis System
- Patient Analysis: Evaluates patient ECG to determine if a rhythm is shockable. Rhythms considered shockable are Ventricular Fibrillation (VF), and certain ventricular tachycardias (VT) associated with lack of circulation. For safety reasons, some VT rhythms associated with circulation will not be interpreted as shockable, and some very low-amplitude or low-frequency rhythms will not be interpreted as shockable VF.
- Sensitivity/Specificity: Meets AAMI DF80 guidelines and AHA recommendations for adult defibrillation (Circulation 1997;95:1677-1682).
- Shock Advised: Able to deliver a shock as soon as the device indicates a shock is advised.
- Quick Shock: Able to deliver a shock after the CPR pause, typically in eight seconds.
- Shock-to-Shock Cycle Time: Typically less than 20 seconds between shocks in a series.
- Artifact Detection: Advanced signal processing allows accurate ECG analysis even in the presence of most pacemaker artifact and electrical noise sources. Other artifacts are detected and corrective voice prompts issued.
- Battery
- Item Number(s): Standard: M5070A, Aviation: 989803139301 (TSO C-142) U.S. only.
- Type: 9 Volt DC, 4.2 Ah, lithium manganese dioxide, disposable long-life primary cell.
- Capacity: Minimum 200 shocks or 4 hours of operating time.
- Install-By-Date: Battery is labeled with an install-by date of at least five years from date of manufacture.
- Standby Life: Four years typical when battery is installed by the install by date. (Will power the AED in standby state within the specified standby temperature range, assuming one battery insertion test and no uses.)
- SMART Pads II
- Item Number: 989803139261
- How Supplied: Disposable pads case containing adhesive multifunction defibrillator pads. HeartStart plug style connector clicks into device for preconnected pads solution.
- Active Surface Area: 2.4 in2 (80 cm2) each
- Cable Length: 48 inches (121.9 cm).
- Use-By-Date: Pads case is labeled with a use-by date of at least two years from date of manufacture.
- Infant/Child Key
- Item Number: 989803139311
- Training Pads II
- Item Number: 989803139271
- Function: Special pads place HeartStart FRx into training mode and disable its energy delivery capability. Features 8 real-world training scenarios.
- Automated and User Activated Self-Tests
- Daily Automatic Self- Tests: Tests internal circuitry, waveform delivery system, pads and battery capacity.
- Pads Integrity Test: Specifically tests readiness-for-use of pads (gel moisture).
- Battery Insertion Test: Upon battery insertion, extensive automatic self-tests and user-interactive test check device readiness.
- Status Indicator: Blinking green "Ready" light indicates ready for use.
- Data Recording and Transmission
- Infrared: Wireless transmission of event data to PC or palmOneTM PDA using the IrDA protocol.
- HeartStart Event Review Software: Data management software (optional) for download and review of data retrieved through defibrillator's infrared data port.
- Data Stored: Store first 15 minutes of ECG and the entire incidents events and analysis decisions.
Request Info
|
|
|
Back |
|
|